The technique of density variation allows for simultaneous determination of size and density distributions. This experiment is feasible for
- obtaining size distributions for particles or molecules of unknown density
- determining particle densities or density distributions
- obtaining size distributions for multimodal mixtures containing species of different density
- systems exhibiting excess properties
- …
The procedure is applicable to both sedimentation velocity and sedimentation equilibrium experiments. It is based on comparative measurements in two or more chemically equivalent solvents of different density – most likely protonated and deuterated variants of the same solvent. The particle or molecule will experience different sedimentation velocity, or bouyant density, respectively, in either solvent. For sedimentation velocity, a density distribution will be obtained; for sedimentation equilibrium it will be a weight average.
The sedimentation velocity variant is rather cheap and quick. Sedimentation velocity is measured in two solvents of different density; combination of the resulting sedimentation coefficient distributions yields
- a particle density distribution and
- a particle size distribution, equivalent to
- a mass distribution
simultanously, where sizes and densities are unambigously assigned to each other. The primary result is a density distribution, allowing for the calculation of sizes and masses under the prerequisite of knowledge on the frictional properties.
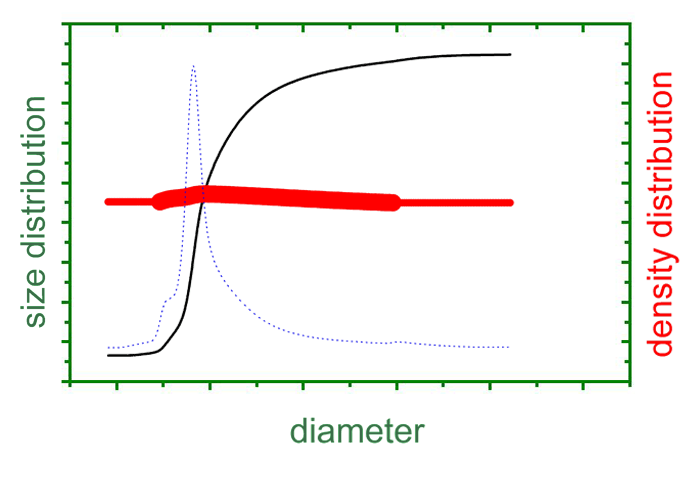
The figure shows the integral and differential particle size distributions in black and blue, respectively. The corresponding densities are plotted in red on the right axis. In this case, a monomodal distribution with nearly constant density is observed.
An interesting aspect in dealing with hard spheres, for example polystyrene latices, is that the diameter obtained is the geometric diameter. From the raw data, the hydrodynamic diameter is obtained. This means that the experiment provides the thickness of the hydration shell.
If the particle is swollen with solvent, the obtained density is the bulk density of the pure material. This applies, for example, for polymer coils and for proteins. For the latter, this means that the most relevant property of hydration is accessible through density variation, a parameter that is most important for understanding the functionality of such molecules.
With increasing complexity of a particle, auxiliary methods must be included for total characterisation. Highly complex aggregates, such as micelles, and probably even exhibiting excess properties, can be fully characterized, yielding parameters such as the diameter of the micelle core or quantifying excess properties.
Concentration and diffusion can take considerable effect on the results of a density variation evaluation, requiring care in defining experimental and evaluation strategies. The technique is most powerful and reliable, taking benefit from the fact that Analytical Ultracentrifugation is the only analytical method based on the fact that accelerated mass exhibits a force – thus targeting on mass and density as the fundamental particle properties. Analytical Ultracentrifugation is the only method capable of measuring particle density distributions.
More theoretical background can be found on our scientific website under www.analytical-ultracentrifugation.com.

